

ECOSHELTA has long been part of the sustainable building revolution and makes high quality architect designed, environmentally minimal impact, prefabricated, modular buildings, using latest technologies. Our state of the art building system has been used for cabins, houses, studios, eco-tourism accommodation and villages. We make beautiful spaces, the applications are endless, the potential exciting.
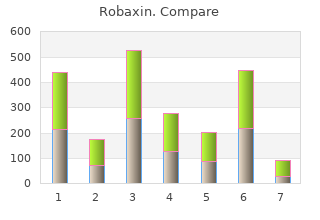
2018, Southwestern Oklahoma State University, Gonzales's review: "Robaxin generic (Methocarbamol) 500 mg. Proven Robaxin.".
Both methods determine joint damage and the progression of radiological damage on continuous scales cheap 500 mg robaxin otc muscle spasms 7 little words. Currently discount robaxin 500 mg without prescription muscle relaxant drugs, no consensus exists on how much progression constitutes a clinically important progression that would have an effect on health outcomes. A re-analysis of published data of 185 patients with early rheumatoid arthritis assessed changes on the modified Sharp score and their association with functional disability (Health 26 Assessment Questionnaire Disability Index). Results indicated that the relation between Sharp score and Health Assessment Questionnaire Disability Index was dependent on the amount of damage (suggesting a threshold effect) and on patients’ age. With lower age, no effect of radiographic joint damage on functional capacity could be demonstrated. Overall a progression of 6 points on the Sharp score was associated with an increase of 0. An international expert panel assessed the minimal clinically important difference in joint damage (from a clinician’s perspective). They used hand and foot radiographs to correlate their findings with the smallest detectable difference on the Sharp/van der Heijde and the Larson/Scott 29 methods. Results suggested that the smallest detectable difference on the Sharp/van der Heijde score reflected a minimal clinically important difference, while the Larson/Scott method was too insensitive to determine relevant changes. This study, however, did not take minimal important differences from a patient perspective into consideration. METHODS Literature Search To identify articles relevant to each key question, for Update 3 we searched PubMed, EMBASE, CINAHL, Centre for Reviews and Dissemination, The Cochrane Library, and International Pharmaceutical Abstracts from 2009 (January) to 2011 (October) using included drugs (abatacept, adalimumab, alefacept, anakinra, certolizumab pegol, etanercept, golimumab, infliximab, natalizumab, rituximab, tocilizumab, and ustekinumab), indications (rheumatoid arthritis, juvenile idiopathic arthritis, ankylosing spondylitis, psoriatic arthritis, Crohn’s disease, ulcerative colitis, and plaque psoriasis), and study designs as search terms (see Appendix B for complete search strategies). We attempted to identify additional studies through hand searches of reference lists of included studies and reviews. In addition, we searched the US Food and Drug Administration Center for Drug Evaluation and Research website for medical and statistical Targeted immune modulators 22 of 195 Final Update 3 Report Drug Effectiveness Review Project reviews of individual drug products. Finally, we requested dossiers of published and unpublished information from the relevant pharmaceutical companies for this review. All received dossiers were screened for studies or data not found through other searches. All citations were imported into an electronic database (Endnote X4, Thomson Reuters). Study Selection Two people independently reviewed abstracts; if both reviewers agreed that the study did not meet eligibility criteria, it was excluded. We obtained the full text of all remaining articles. Records were considered for exclusion if they did not meet pre-established eligibility criteria with respect to study design or duration, patient population, interventions, outcomes, and comparisons to medications outside our scope of interest. With respect to study design we took a “best evidence” approach for this review. Results from well-conducted, head-to-head trials provide the strongest evidence to compare drugs with respect to effectiveness, efficacy, and adverse events; head-to-head trials were defined as those comparing one targeted immune modulator with another. Randomized controlled trials of at least 12 weeks duration with an outpatient study population were eligible for inclusion. In addition, we reviewed well-conducted, head-to-head observational studies with a follow-up of at least 12 weeks to augment findings from experimental studies. Long-term observational studies can provide evidence on outcomes that may be difficult to observe in randomized controlled trials due to limitations in sample sizes and study durations. Furthermore, observational data can provide information whether treatment effects observed in randomized 30 controlled trials can be translated to less selected populations. Nevertheless, the strength of evidence of these results for comparing different drugs must be rated lower than results from the most preferred type of trial. If no head-to-head evidence was published, we reviewed placebo-controlled trials for indications of interest. We reviewed all placebo-controlled trials to provide an overview of efficacy without taking drug equivalency into account. Study populations, disease severity, and concomitant treatments can differ considerably across placebo-controlled trials. Comparisons of treatment effects across trials must, therefore, be made with caution. We included meta-analyses in the evidence report if they were relevant to a key question 31 and of good or fair methodological quality. For each section, we included results from the most recent and best-quality systematic review and meta-analysis and did not include data from older meta-analyses where these had been superseded in terms of included studies and analysis.
Response is typically migrated from the initial trials’ threshold of molecular relapse “biphasic” (rapid decline followed by slower decline) and in a sense (detectable disease generally in the 4- to 4 cheap robaxin 500 mg online muscle relaxer 86 67. Although buy robaxin 500 mg with amex spasms 1983 download, at face value, this may portend that “failure” of as demonstrated by the preliminary data of ponatinib’s activity in the experiment of cessation may carry with it a 1. Therefore, as long as early milestones are met, therein, several studies have addressed the pragmatic reality of declining disease is acceptable. As noted above, caution is advised patients’ MRD, its stability, and consideration for TFR trials. It in calling response “failure” too early, such as in the case of may be that the stability of deep remission may have little to do seemingly inadequate early transcript reduction based on black and with the success of treatment cessation; in the According to white thresholds (10% IS at 3 months), because individualization of STIM (A-STIM) trial, patients who discontinued from a stable response and accounting for initial disease burden may change CMR and an ‘unstable CMR’ had equal chances of success. Long-term prognostic significance of early molecular response to imatinib in newly diagnosed In essence, disease burden in CML is too much if it is permissive chronic myeloid leukemia: an analysis from the International Random- ized Study of Interferon and STI571 (IRIS). Increasing understanding into the nature of stem cell transcripts in chronic myeloid leukaemia patients in complete remission biology and leukemogenesis, described by many as an accumula- using the polymerase chain reaction and nested primers. Absolute quantitative focus away from the quantity of residual disease but more on the detection of ABL tyrosine kinase domain point mutations in chronic quality of the remission. It may be that the benefit of better CML myeloid leukemia using a novel nanofluidic platform and mutation- therapy is a result, not of eliminating more of it, but of specific PCR. Molecular remission in chronic doing so expeditiously. It is unsettling to patients and dissatisfy- myeloid leukemia patients with sustained complete cytogenetic remis- ing to practitioners, but it appears that the “little” disease sion after imatinib mesylate treatment. BCR-ABL messenger RNA too much as stable responses are the expectation and treatment- levels continue to decline in patients with chronic phase chronic free remissions appear to be a potential reality for many. European LeukemiaNet recommendations for the management of chronic myeloid leukemia: Disclosures 2013. Conflict-of-interest disclosure: The author has consulted for Ariad, 16. International randomized Pfizer, Bristol Myers Squibb, and Novartis Oncology. Off-label study of interferon vs STI571 (IRIS) 8-year follow up: sustained drug use: None disclosed. Mauro, MD, Memorial Sloan Kettering Cancer Center, 17. Effects of a selective response to imatinib predicts cytogenetic and clinical outcome in inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive chronic myeloid leukaemia. Efficacy and safety of a specific levels at 3 months is the only requirement for predicting outcome for inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. Hanfstein B, Mu¨ller MC, Hehlmann R, et al; SAKK; German CML interferon and low-dose cytarabine for newly diagnosed chronic-phase Study Group. Early molecular and cytogenetic response is predictive for chronic myeloid leukemia. Italian Cooperative Study Group on Chronic Myeloid Leukemia. Interferon alfa-2a as compared with conventional chemotherapy for the 21. Assessment at 6 months may be treatment of chronic myeloid leukemia. Prognosis for patients with competitive polymerase chain reaction. CML and 10% Bcr-Abl1 after 3 months of imatinib depends on the 7. Velocity of early myelogenous leukemia are associated with excellent long-term progno- BCR-ABL transcript elimination as an optimized predictor of outcome sis. Predictive value of early molecular alternative to bone marrow cytogenetics. Bru¨mmendorf TH, Kantarjian HM, Gambacorti-Passerini C, et al. Discontinuation of imatinib in patients with clinical outcomes in the phase 3 BELA study [abstract]. Blood (ASH chronic myeloid leukaemia who have maintained complete molecular Annual Meeting Abstracts).
Interpreting the clinical importance of group differences in chronic pain clinical trials: IMMPACT recommendations buy generic robaxin 500 mg online muscle relaxant 303. Research design considerations for confirmatory chronic pain clinical trials: IMMPACT recommendations robaxin 500mg sale quetiapine spasms. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. Core outcome domains for chronic pain clinical trials: IMMPACT recommendations. Initiative on Methods, Measurement and Pain Assessment in Clinical Trials. Developing patient-reported outcome measures for pain clinical trials: IMMPACT recommendations. Analyzing multiple endpoints in clinical trials of pain treatments: IMMPACT recommendations. Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials. Identifying important outcome domains for chronic pain clinical trials: an IMMPACT survey of people with pain. Elbourne DR, Altman DG, Higgins JP, Curtin F, Worthington HV, Vail A. Meta-analyses involving cross-over trials: methodological issues. Validation of an index of the quality of review articles. Grading the strength of a body of evidence when comparing medical interventions. Methods Guide for Comparative Effectiveness Reviews. Grading the strength of a body of evidence when comparing medical interventions-Agency for Healthcare Research and Quality and the Effective Health Care Program. The results of direct and indirect treatment comparisons in meta-analysis of randomized controlled trials. The PRISMA statement for reporting systematic reviews and emta-analyses of studies that evaluate health care interventions: explanation and elaboration. Neuropathic pain 52 of 92 Final Update 1 Report Drug Effectiveness Review Project 37. Bansal D, Bhansali A, Hota D, Chakrabarti A, Dutta P. Randomized double-blind study comparing the efficacy and safety of lamotrigine and amitriptyline in painful diabetic neuropathy. Central post-stroke pain--a controlled trial of amitriptyline and carbamazepine. A Placebo-Controlled Trial of Pregabalin and Amitriptyline for Treatment of Painful Diabetic Peripheral Neuropathy. Comparison of the effectiveness of amitriptyline and gabapentin on chronic neuropathic pain in persons with spinal cord injury. Morello CM, Leckband SG, Stoner CP, Moorhouse DF, Sahagian GA. Randomized double-blind study comparing the efficacy of gabapentin with amitriptyline on diabetic peripheral neuropathy pain. Nortriptyline and gabapentin, alone and in combination for neuropathic pain: a double-blind, randomised controlled crossover trial. Gabapentin versus nortriptyline in post-herpetic neuralgia patients: a randomized, double-blind clinical trial--the GONIP Trial. International Journal of Clinical Pharmacology & Therapeutics. Low-dose gabapentin as useful adjuvant to opioids for neuropathic cancer pain when combined with low-dose imipramine.
Alloimmunization remains a chronic transfusions and maintaining a hemoglobin S percentage of major complication associated with RBC transfusions in patients less than 30% generic 500 mg robaxin otc muscle relaxant herbs. Allo- and sions has led to recurrent stroke generic robaxin 500 mg free shipping spasms stomach, even with transition to hy- auto-antibodies increase the complexity of compatibility testing and droxyurea. This review with their first stroke appear to be at highest risk for recurrent stroke summarizes the main indications for RBC therapy for SCD, despite maintaining maximum hemoglobin S levels less than 50% focusing specifically on the immunohematological complications or even 30%. Transfusion-associated iron overload is the subject of an still at risk for silent cerebral infarcts5,7 and cerebral vasculopathy accompanying chapter. Acute infarctive stroke is A recent randomized phase 3 trial, Stroke With Transfusions managed with RBC transfusion to reduce the percent hemoglobin S Changing to Hydroxyurea (SWiTCH), addressed transition to level to below 30% to prevent progression of cerebral ischemia. The hydroxyurea therapy for patients with a history of stroke and iron optimal transfusion method to provide the best neurologic outcome overload. One multi-institutional retrospective study found that with iron chelation), there was no difference in liver iron content for exchange transfusion was associated with a lower risk of subsequent patients treated with hydroxyurea and phlebotomy. Major clinical trials addressing transfusion indications for SCD Major clinical trials addressing transfusion indications for SCD Primary stroke prevention Stroke Prevention Study in Sickle Cell Anemia (STOP): Showed that prophylactic chronic transfusion prevents stroke in children with SCD who have abnormal transcranial Doppler (TCD) ultrasounds Stroke Prevention Study in Sickle Cell Anemia 2 (STOP 2): Showed that discontinuation of chronic transfusion results in an increased rate of abnormal TCD conversion and overt stroke TCDs With Transfusions Changing to Hydroxyurea (TWITCH)*: Compares hydroxyurea against transfusions for children who currently receive chronic transfusion therapy to reduce the risk of primary stroke Secondary stroke prevention Stroke With Transfusions Changing to Hydroxyurea (SWiTCH): Showed that transfusion and chelation remain the optimal choice for prevention of stroke recurrence and iron overload Silent infarcts Silent Infarct Transfusion Trial (SIT)†: Determine the effectiveness of blood transfusion therapy for the prevention or progression of silent cerebral infarcts Acute chest syndrome, acute National Acute Chest Syndrome Study Group: Showed that transfusion improves oxygenation Preoperative management The Preoperative Transfusion in Sickle Cell Disease Study Group: Showed that a conservative transfusion regimen is as effective as an aggressive regimen in preventing perioperative complications Transfusion Alternatives Preoperatively in Sickle Cell Disease (TAPS): Showed that preoperative transfusion is associated with decreased perioperative complications *Currentlyrecruitingsubjects. At the magnetic resonance angiography–defined vasculopathy but normal time of study closure, no strokes were documented in patients TCD velocities would benefit from RBC transfusions to prevent receiving transfusions with chelation, but 7 patients (10%) receiving vasculopathy progression and/or silent or overt strokes. Young hydroxyurea and phlebotomy had experienced a new stroke. BABY HUG trial identified no patients at a mean age of 12. The Stroke Prevention Study in Sickle Cell Disease (STOP) demonstrated a Acute chest syndrome 92% stroke risk reduction among 63 of 130 children with abnormal Acute chest syndrome (ACS) describes a new pulmonary infiltrate TCD results. ACS is the leading cause of death for at-risk patients has been universally adopted. The management of ACS is primarily supportive nitely because discontinuation resulted in an increased rate of and includes respiratory therapy, antibiotics, and, often, RBC abnormal TCD conversion and overt stroke. There have been no randomized controlled trials fusions on the STOP 2 trial was also associated with a higher comparing either simple or exchange transfusion versus no transfu- occurrence of silent cerebral infarcts, documented in 3 of 37 patients sion in patients with SCD and ACS. One small study has shown son of hydroxyurea and transfusion therapy for children with that simple transfusion is equally effective for the transfusion abnormally elevated TCD velocities but no primary stroke is management of ACS as exchange transfusion. The rationale is based on previous studies demon- sive respiratory decline or persistent hypoxia despite simple strating that hydroxyurea can lower TCD velocities in patients with transfusion. One small prospective study demonstrated with these lesions at baseline and with normal TCD velocities. Hydroxyurea use is associated with lower rates of tions in patients with SCD, with 13 of 33 (39%) patients in the no ACS26 and is indicated for prevention of recurrent ACS. However, the TAPS trial was not able to rocytapheresis after failure to respond to simple transfusions. A address 2 major questions, what is the best preoperative manage- dramatic reduction in hospitalization for ACS (and pain) was ment of individuals with other subtypes of SCD (HbSC or S observed in children undergoing chronic transfusion for primary thalassemia) and what is the optimal management for low-risk stroke prevention compared with the observed group. Pulmonary hyperten- occurring with acute splenic sequestration and transient RBC sion affects up to 30% of adults with SCD and strongly predicts morbidity,34 but there is no proven treatment. Acute splenic sequestration is typically accompanied by a precipitous decrease in hemoglobin level and the potential for transfusions and long-term anticoagulation have been suggested but hypovolemic shock. The immediate treatment is directed toward require investigation. Pregnant women with SCD have increased correction of hypovolemia with RBC transfusion. Because RBCs maternal and fetal mortality and morbidity. RBC transfusion is sequestered in the spleen are remobilized, patients should be indicated for the treatment of acute complications during pregnancy, transfused cautiously to prevent hyperviscosity after splenic seques- but there are insufficient data currently to recommend its use prophylactically. Aliquots of 5 mL/kg may be administered, along with close monitoring of the spleen size, hemoglobin level, and determine the effectiveness of transfusion therapy for acute manage- cardiovascular status of the child. In cases of severe sequestration ment or prevention of vasoocclusive painful episodes, priapism, or and anemia with hypovolemic shock, initial transfusion with 10 prevention of other end organ damage due to SCD. In a retrospective multicenter study of 190 patients with SCD of genotypes SS or S 0, 67% of infants with Complications of transfusion therapy splenic sequestration had one or more recurrent episodes.